-
Centers for Disease Control/Division of Parasitic Diseases and Malaria
EOL staff
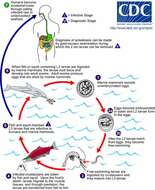
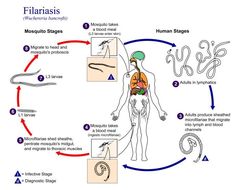
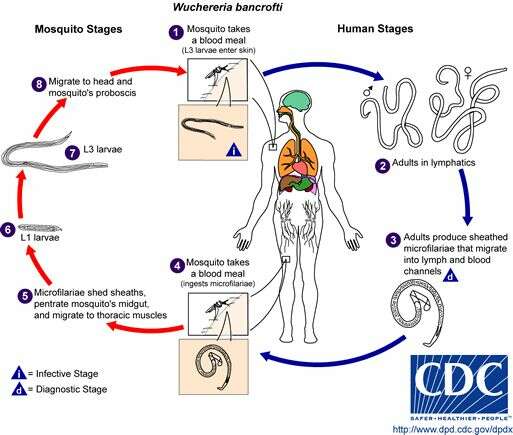
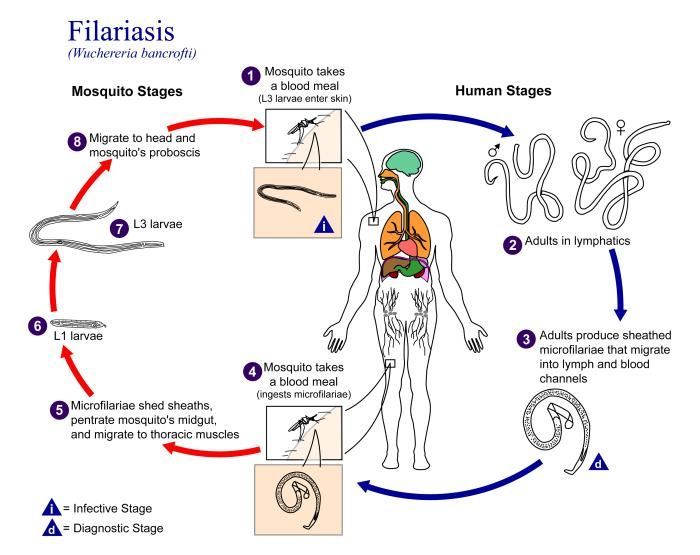
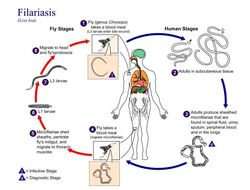
Life cycle of Wuchereria bancroftiVarious species of mosquitoes are known vectors of Wuchereria bancrofti filariasis, depending on geographic distribution. Among these are: Culex (C. annulirostris, C. bitaeniorhynchus, C. quinquefasciatus, and C. pipiens); Anopheles (A. arabinensis, A. bancroftii, A. farauti, A. funestus, A. gambiae, A. koliensis, A. melas, A. merus, A. punctulatus and A. wellcomei); Aedes (A. aegypti, A. aquasalis, A. bellator, A. cooki, A. darlingi, A. kochi, A. polynesiensis, A. pseudoscutellaris, A. rotumae, A. scapularis, and A. vigilax); Mansonia (M. pseudotitillans, M. uniformis); and Coquillettidia (C. juxtamansonia). During a blood meal, an infected mosquito introduces third-stage filarial larvae onto the skin of the human host, where they penetrate into the bite wound (1). They develop in adults that commonly reside in the lymphatics (2). The female worms measure 80 to 100 mm in length and 0.24 to 0.30 mm in diameter, while the males measure about 40 mm by 0.1 mm. Adults produce microfilariae measuring 244 to 296 μm by 7.5 to 10 μm, which are sheathed and have nocturnal periodicity, except for the South Pacific microfilariae, which lack marked periodicity. The microfilariae migrate into lymph and blood channels moving actively through lymph and blood (3). A mosquito ingests the microfilariae during a blood meal (4). After ingestion, the microfilariae lose their sheaths and some of them work their way through the wall of the proventriculus and cardiac portion of the mosquito's midgut and reach the thoracic muscles (5). There the microfilariae develop into first-stage larvae (6) and subsequently into third-stage infective larvae (7). The third-stage infective larvae migrate through the hemocoel to the mosquito's prosbocis (8) and can infect another human when the mosquito takes a blood meal (1).From
Centers for Disease Control Parasites and Health website.
-
Saša Širca, Gregor Urek, Stela Lazarova, Milka Elshishka, Vlada Peneva
Zookeys
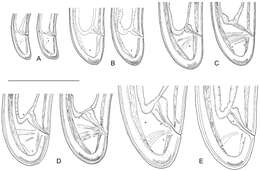
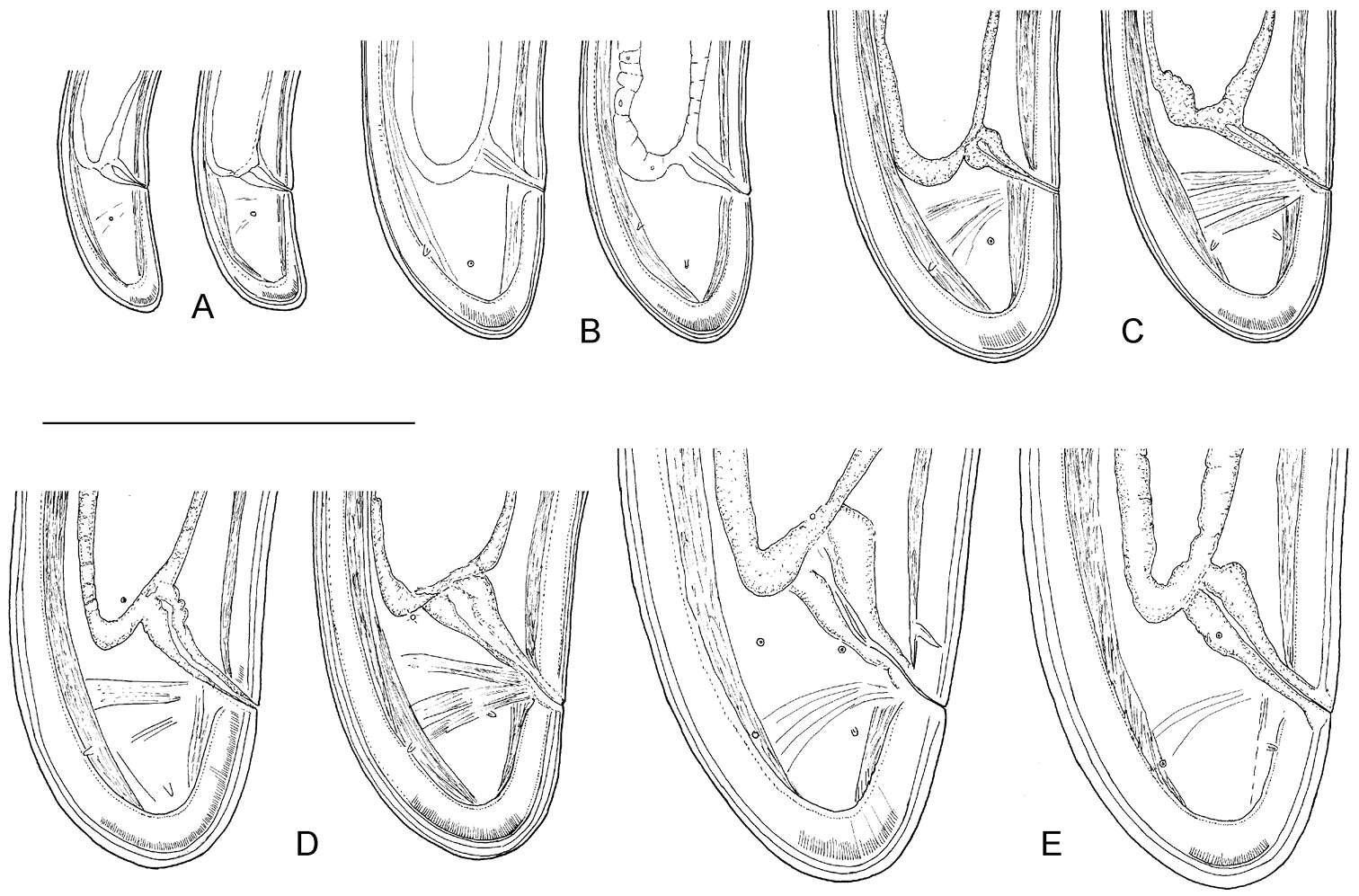
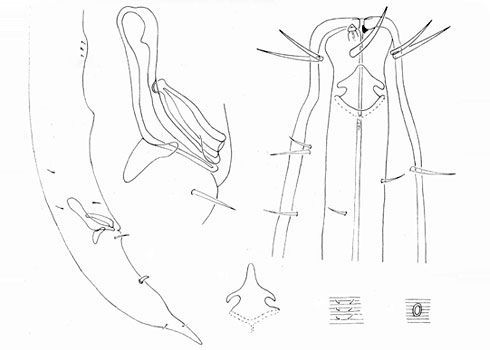
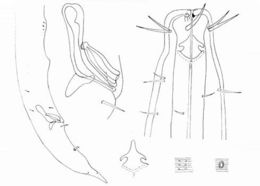
Figure 9.Longidorus carniolensis sp. n. Evolution of the tail. A–D Tail of first–fourth juvenile stage E Tail of female. Scale bar: 100 μm.
-
Centers for Disease Control/Division of Parasitic Diseases and Malaria
EOL staff
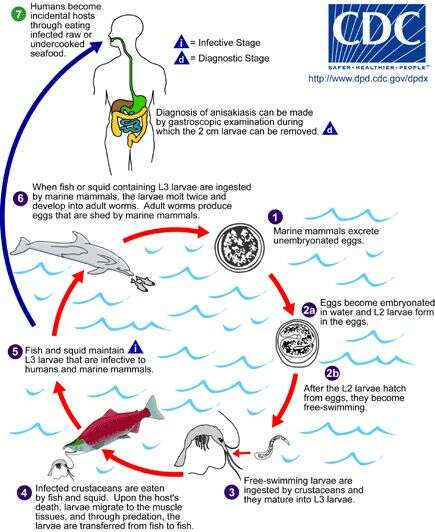
Life cycle of Anisakis simplex and Pseudoterranova decipiens, parasitic agents of anisakiasis in humansAdult stages of Anisakis simplex and Pseudoterranova decipiens reside in the stomachs of marine mammals, where they are embedded in the mucosa in clusters. Unembryonated eggs produced by adult females are passed in the feces of marine mammals (1). The eggs become embryonated in water, and first-stage larvae are formed in the eggs. The larvae molt, becoming second-stage larvae (2a), and after the larvae hatch from the eggs, they become free-swimming (2b). Larvae released from the eggs are ingested by crustaceans (3). The ingested larvae develop into third-stage larvae that are infective to fish and squid (4). The larvae migrate from the intestine to the tissues in the peritoneal cavity and grow up to 3 cm in length. Upon the host's death, larvae migrate to the muscle tissues, and through predation, the larvae are transferred from fish to fish. Fish and squid maintain third-stage larvae that are infective to humans and marine mammals (5). When fish or squid containing third-stage larvae are ingested by marine mammals, the larvae molt twice and develop into adult worms. The adult females produce eggs that are shed by marine mammals (6). Humans become infected by eating raw or undercooked infected marine fish (7). After ingestion, the anisakid larvae penetrate the gastric and intestinal mucosa, causing the symptoms of anisakiasis.From
Centers for Disease Control Parasites and Health website.
-
Centers for Disease Control/Division of Parasitic Diseases and Malaria
EOL staff
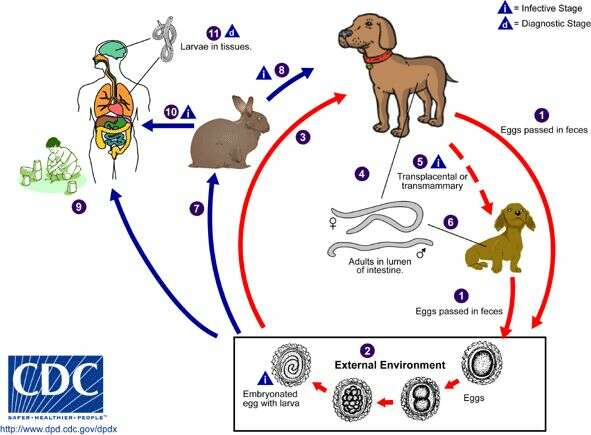
Life cycle of the nematode (roundworm) Toxocara canis, the most common cause of Toxocariasis in humansToxocara canis completes its life cycle in dogs, with humans acquiring the infection as accidental hosts. Unembryonated eggs are shed in the feces of the definitive host (1). Eggs embryonate and become infective in the environment (2). Following ingestion by dogs (3), the infective eggs hatch and larvae penetrate the gut wall. In younger dogs, the larvae migrate through the lungs, bronchial tree, and esophagus; adult worms develop and oviposit in the small intestine (4). In older dogs, patent infections can also occur, but larval encystment in tissues is more common (an infection is "patent" when direct evidence of the organism can be detected, e.g., in the patient’s feces or blood, regardless of whether symptoms have appeared). Encysted stages are reactivated in female dogs during late pregnancy and infect by the transplacental and transmammary routes the puppies (5), in whose small intestine adult worms become established (6). Puppies are a major source of environmental egg contamination. Toxocara canis can also be transmitted through ingestion of paratenic hosts ("transport hosts"): eggs ingested by small mammals (e.g. rabbits) hatch and larvae penetrate the gut wall and migrate into various tissues where they encyst (7). The life cycle is completed when dogs eat these hosts (8) and the larvae develop into egg-laying adult worms in the small intestine. Humans are accidental hosts who become infected by ingesting infective eggs in contaminated soil (9) or infected paratenic hosts (10). After ingestion, the eggs hatch and larvae penetrate the intestinal wall and are carried by the circulation to a wide variety of tissues (liver, heart, lungs, brain, muscle, eyes) (11). While the larvae do not undergo any further development in these sites, they can cause severe local reactions that are the basis of toxocariasis. The two main clinical presentations of toxocariasis are visceral larva migrans and ocular larva migrans. Diagnosis is usually made by serology or the finding of larvae in biopsy or autopsy specimens.From
Centers for Disease Control Parasites and Health website.
-
Centers for Disease Control/Division of Parasitic Diseases and Malaria
EOL staff
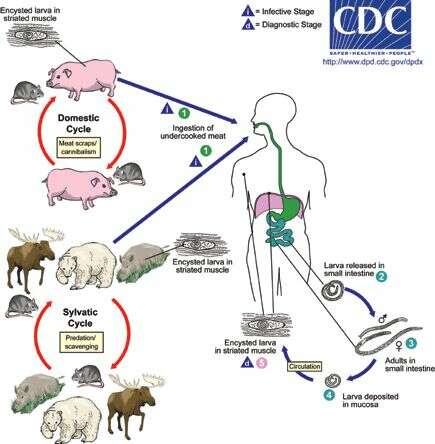
Life cycle of Trichinella nematodes (roundworms), the cause of trichinellosis (trichinosis) in humansDepending on the classification used, there are several species of Trichinella (the nematode parasites responsible for
trichinellosis): T. spiralis, T. pseudospiralis, T. nativa, T. murelli, T. nelsoni, T. britovi, T. papuae, and T. zimbabwensis, all but the last of which have been implicated in human disease. Adult worms and encysted larvae develop within a single vertebrate host and an infected animal serves as a definitive host and potential intermediate host. A second host is required to perpetuate the life cycle. The domestic cycle most often involved pigs and anthropophilic rodents, but other domestic animals such as horses can be involved. In the sylvatic cycle, the range of infected animals is great, but animals most often associated as sources of human infection are bear, moose, and wild boar. Trichinellosis is caused by the ingestion of undercooked meat containing encysted larvae (except for T. pseudospiralis and T. papuae, which do not encyst) of Trichinella species (1). After exposure to gastric acid and pepsin, the larvae are released from the cysts (2) and invade the small bowel mucosa where they develop into adult worms (3). Females are 2.2 mm in length; males 1.2 mm. The life span in the small bowel is about four weeks. After 1 week, the females release larvae (4) that migrate to striated muscles, where they encyst (5). Diagnosis is usually made based on clinical symptoms and is confirmed by serology or identification of encysted or non-encysted larvae in biopsy or autopsy specimens.From
Centers for Disease Control Parasites and Health website.
-
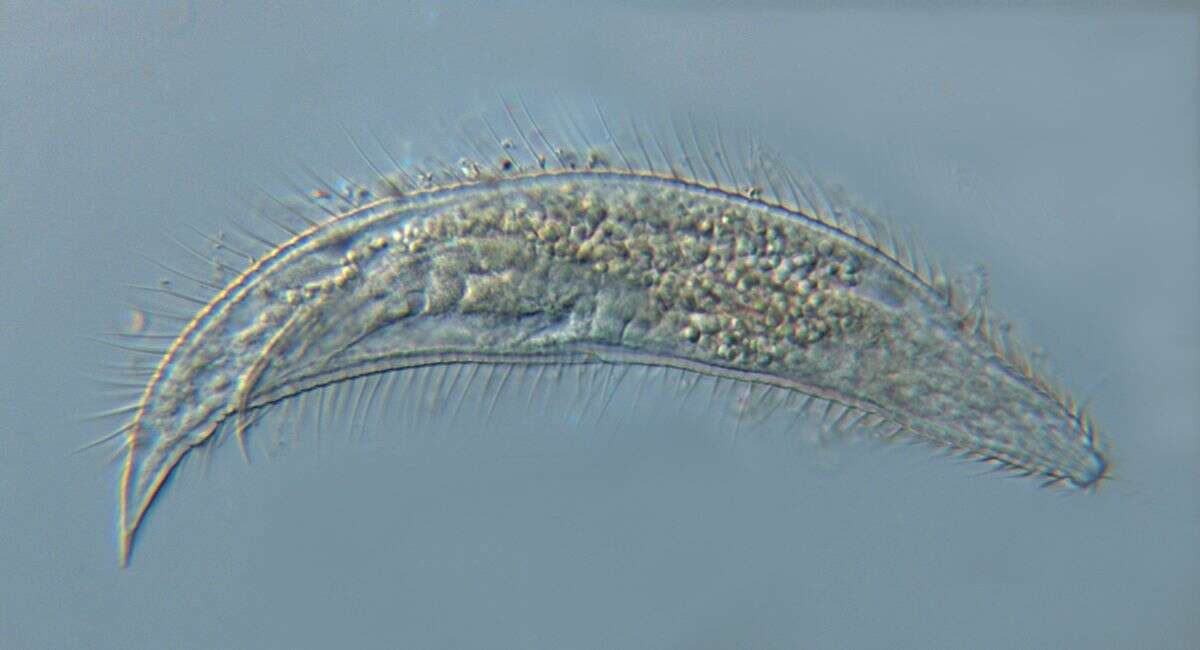
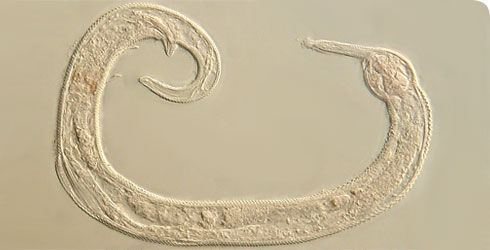
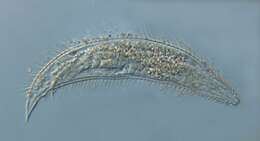
Greeffiella (undescribed species) under approx. 1000x magnification
-
This figure was first published in 1966, describing Platycoma sudafricana from South Africa (Inglis, 1966).
-
The marine nematode worm Manunema kithara was discovered in Kuwait Bay in the northern Arabian Gulf.
-
Magnified 125X, this photomicrograph revealed the presence of a fertile Ascaris sp. egg that was found in an unstained formalin-preserved stool sample. See PHIL 411 for an example of an unfertilized Ascaris lumbricoides egg.Geographic Distribution:The most common human helminthic infection, Ascaris sp. have a worldwide distribution. Their highest prevalence is in tropical and subtropical regions, and areas with inadequate sanitation. Ascariasis occurs in rural areas of the southeastern United States.Created: 1973
-
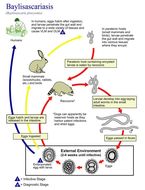
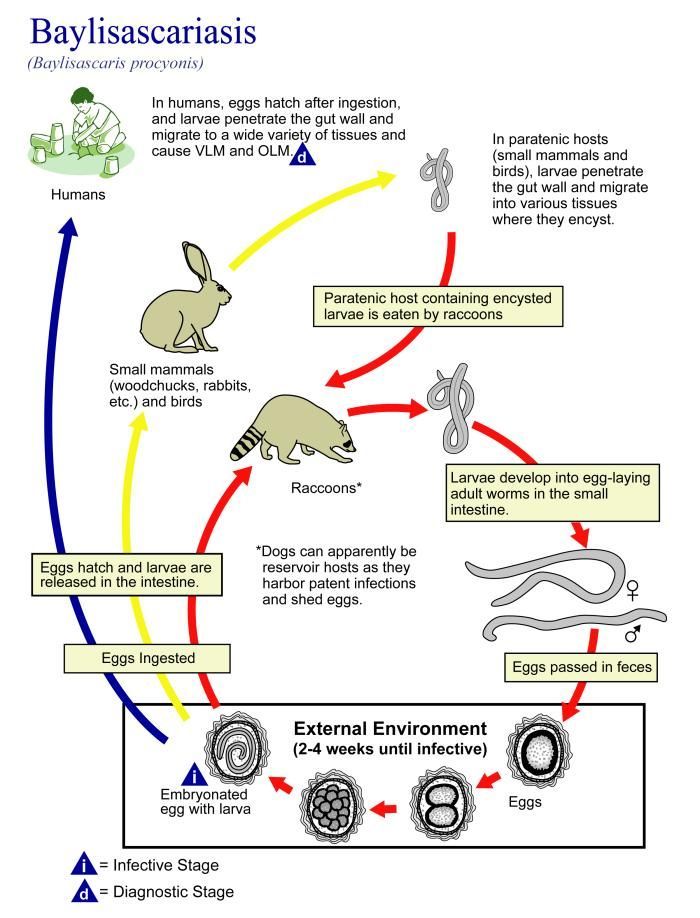
This illustration depicts the life cycle of Baylisascaris procyonis, the causal agent of Baylisascariasis.Created: 2002
-
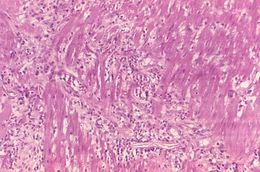
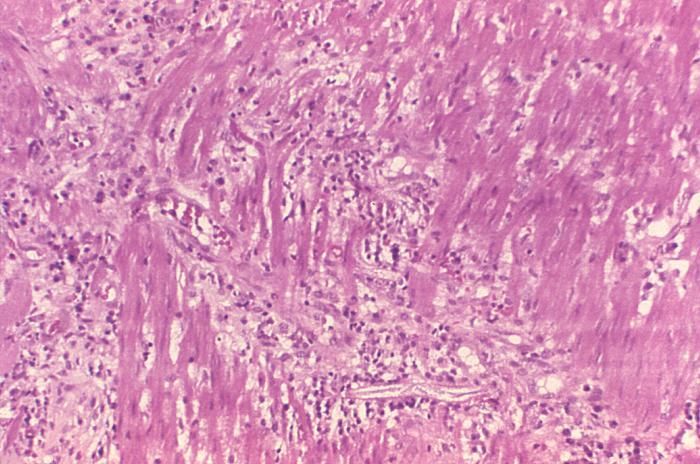
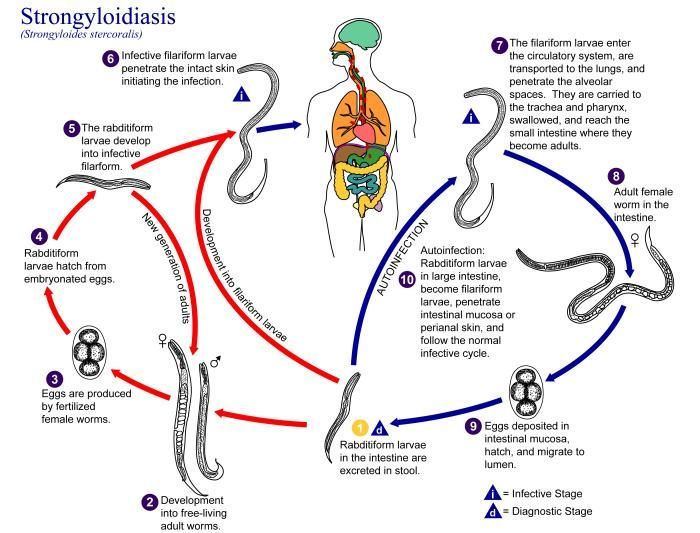
This photomicrograph reveals histopathologic changes indicative of the presence of the intestinal parasitic nematode (roundworm), Strongyloides stercoralis.Clinical Features:Frequently asymptomatic. Gastrointestinal symptoms include abdominal pain and diarrhea. Pulmonary symptoms (including Loefflers syndrome) can occur during pulmonary migration of the filariform larvae. Dermatologic manifestations include urticarial rashes in the buttocks and waist areas. Disseminated strongyloidiasis occurs in immunosuppressed patients, can present with abdominal pain, distension, shock, pulmonary and neurologic complications and septicemia, and is potentially fatal. Blood eosinophilia is generally present during the acute and chronic stages, but may be absent with dissemination.Created: 1972
-
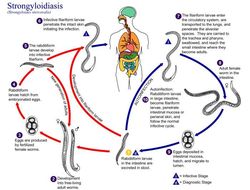
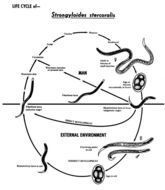
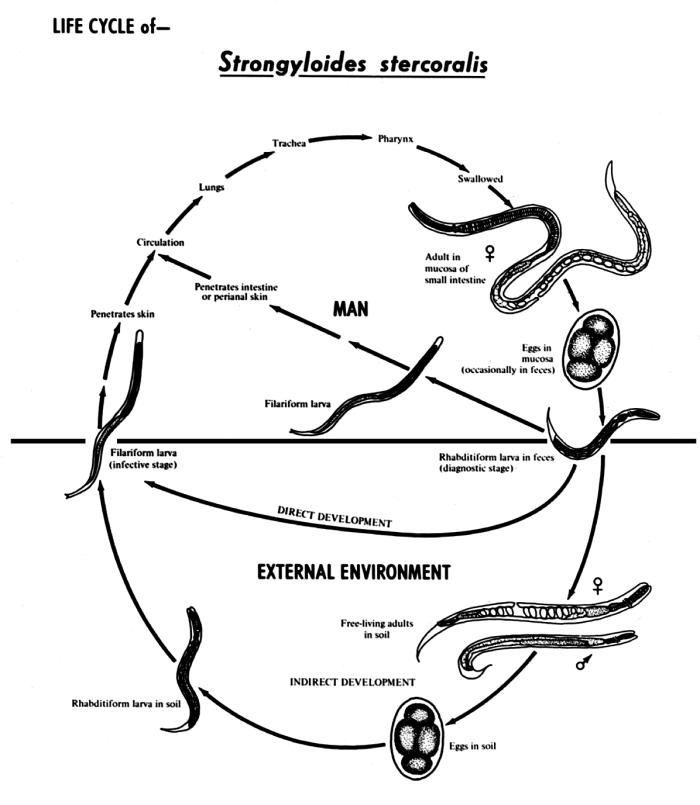
This is an illustration of the life cycle of Strongyloides stercoralis, the causal agent of Strongyloidiasis.Created: 2002
-
This diagram depicts the various stages in the life cycle of the Strongyloides stercoralis nematode.Created: 1982
-
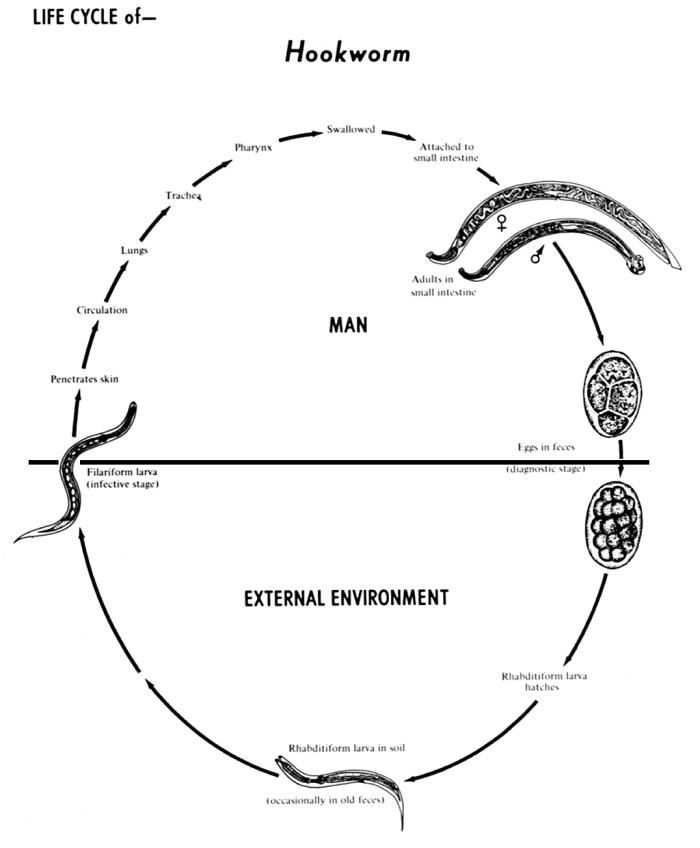
The human hookworms include two nematode (roundworm) species, Ancylostoma duodenale and Necator americanus.Created: 1982
-
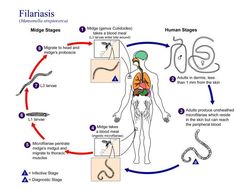
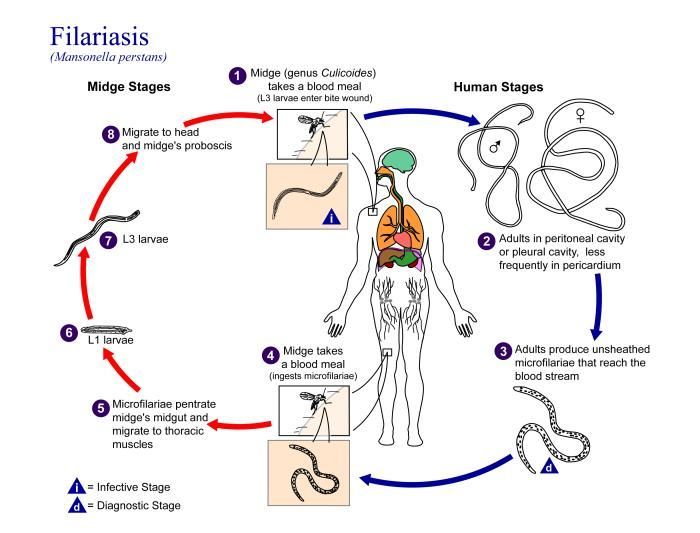
This is an illustration of the life cycle of Mansonella perstans, one of the causal agents of Filariasis.Created: 2002
-
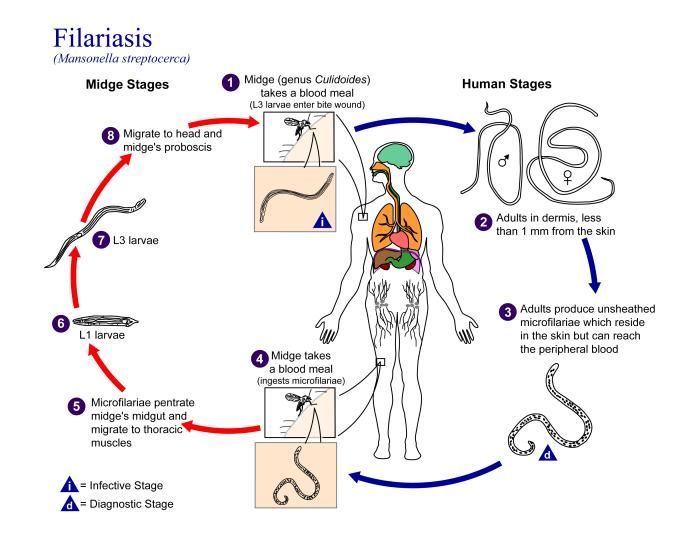
This is an illustration of the life cycle of Mansonella streptocerca, one of the causal agents of Filariasis.Created: 2002
-
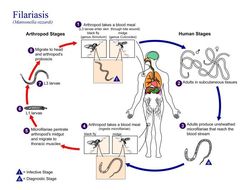
This is an illustration of the life cycle of Wuchereria bancrofti, one the causal agents of Filariasis.Created: 2003
-
This is an illustration of the life cycle of Loa loa, one of the causal agents of Filariasis.Created: 2003
-
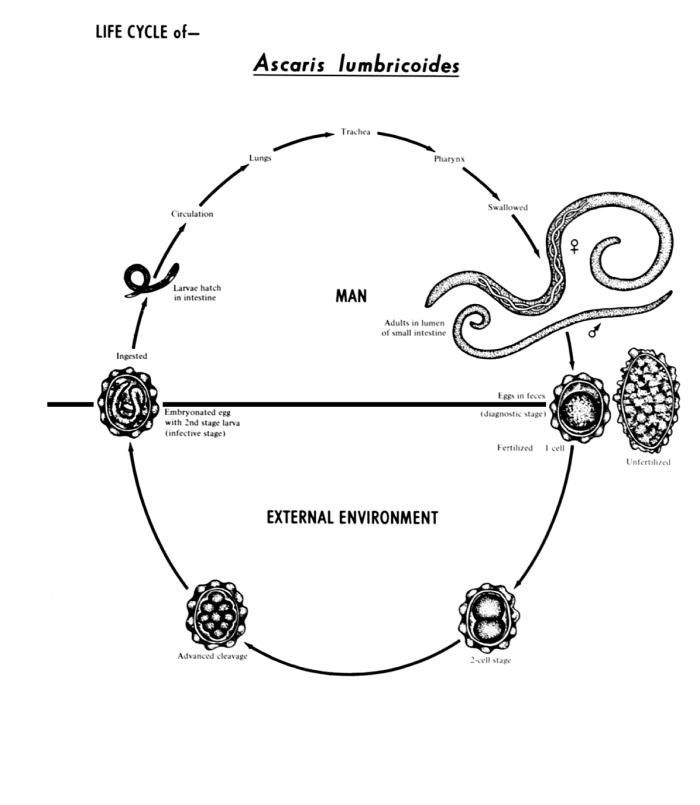
This diagram depicts the various stages in the life cycle of the intestinal roundworm nematode Ascaris lumbricoides.Created: 1982
-
Depicted in this 1960 photograph were two Ascaris lumbricoides nematods, i.e., roundworms. The larger of the two was the female of the species, while the normally smaller male was on the right. Adult female worms can grow over 12 inches in length.Created: 1960
-
This 2007 photograph depicted Center for Disease Control/ NCZVED/DPD laboratory technician, Henry Bishop holding a mass of Ascaris lumbricoides worms, which had been passed by a child in Kenya, Africa. This nematode parasitizes the human small intestine, and is spread from human to human by the fecal-oral route. Children seem to be infected more often than adults, and though the organisms depicted here originated in Africa, the disease can be acquired in the southeastern United States.Created: 2007
-
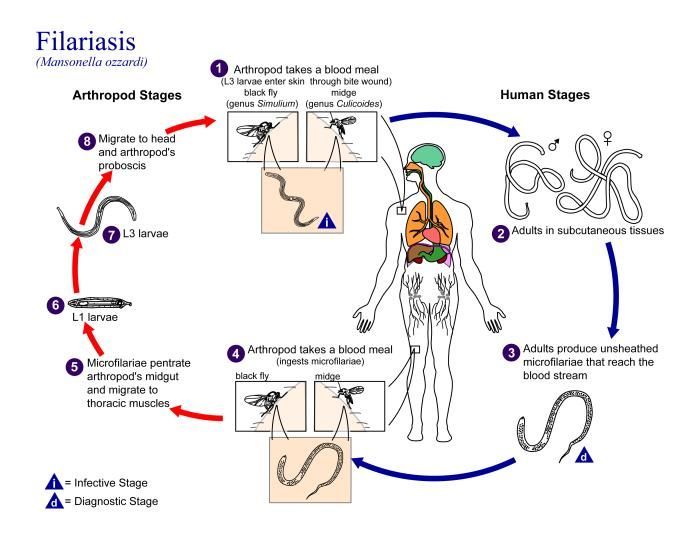
This is an illustration of the life cycle of Mansonella ozzardi, one of the causal agents of Filariasis.Created: 2002
-
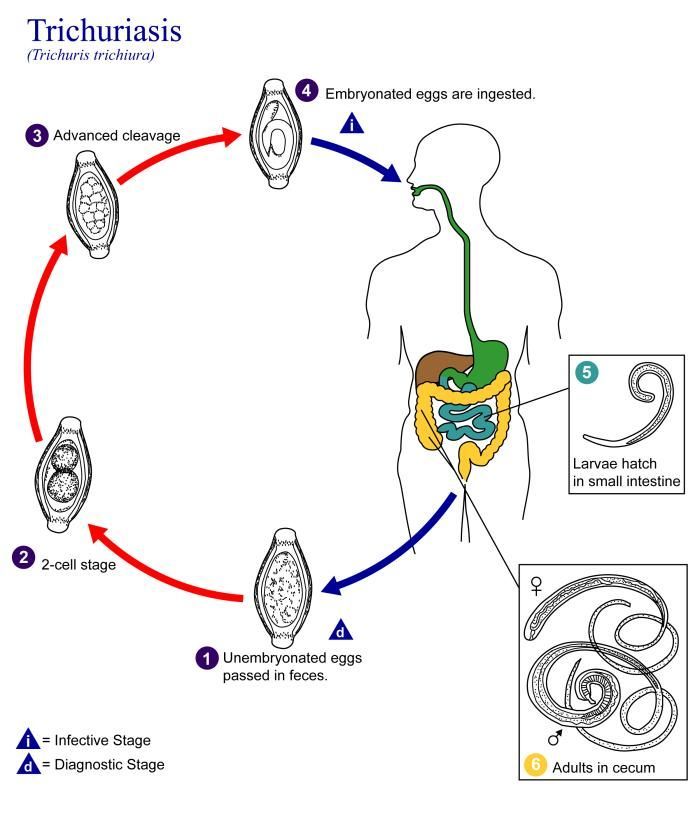
This is an illustration of the life cycle of Trichuris trichiura, the causal agent of Trichuriasis.Created: 2002
-
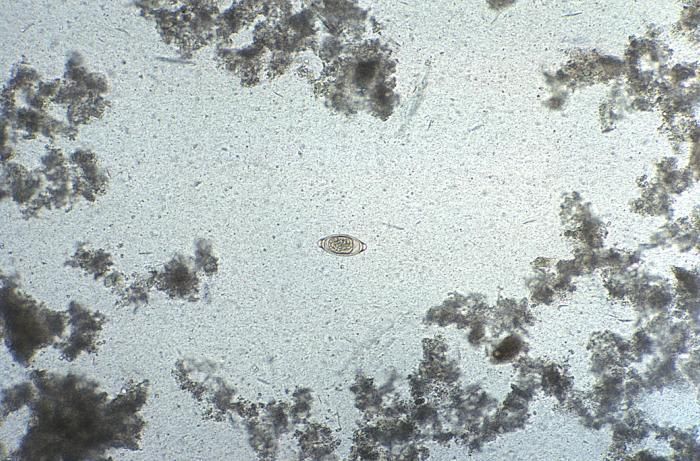
Magnified 128X, this photomicrograph revealed some of the ultrastructural features displayed by a Trichuris trichiura egg. The nematode, or roundworm, is also called the human whipworm. T. trichiura eggs are 50µm-55µm by 20µm-25µm. As is exemplified by this specimen, eggs are football-shaped, thick-shelled, and possess a pair of polar plugs at each end. Eggs are passed unembryonated in stool.Created: 1979